More Pediatricians Prescribe Natroba™ (Spinosad)
Natroba™ (Spinosad) is a proven safe and effective solution for resistant head lice. To date, more than 80% of pediatric specialists in the U.S. have treated head lice with the active compound Spinosad.1,5
Superiority to Nix®
Natroba™ (Spinosad) is the only FDA-approved prescription head lice treatment that demonstrated clinical superiority to Nix® (permethrin 1%) in two head-to-head, phase III clinical trials. No nit combing required.2
In two multicenter, randomized, controlled clinical studies under actual-use conditions, significantly more patients using Natroba™ (Spinosad) were lice-free (no live lice, adults or nymphs) 14 days after the last treatment without nit combing compared to permethrin with combing, 84.6% vs. 44.9% and 86.7% vs. 42.9%, respectively (P<0.001).2
Soil microorganism at the center of the safety profile
The active ingredient in Natroba™ (Spinosad) derives from a naturally occurring soil microorganism3 that differs from agents such as permethrins and pyrethrins4:
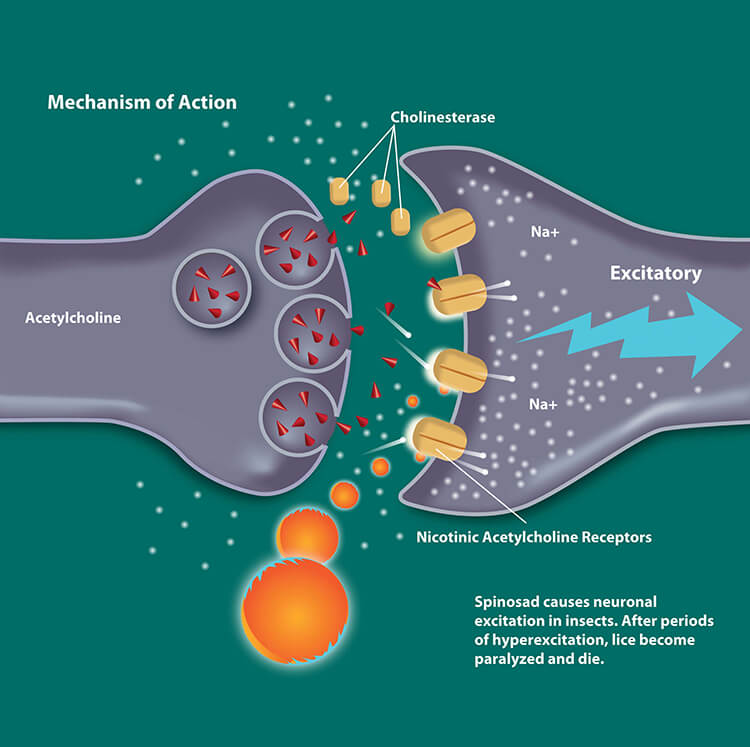
- The active compound, Spinosad, is not systemically absorbed, even in patients as young as six (6) months of age5
- Spinosad targets lice and nits where they live, not penetrating beyond the stratum corneum, before sloughing off through the natural process of non-pathologic desquamation (over approximately 14 days)2
- In a Phase I clinical trial, fourteen (14) subjects 4-15 years of age, applied a single topical (scalp) treatment of Spinosad 1.8% (double the active amount of compound in Natroba™) for 10 minutes. Results demonstrated no systemic absorption of Spinosad2
- There is no evidence of neurotoxicity, developmental/reproductive toxicity, immunotoxicity, mutagenicity, or carcinogenicity from Spinosad exposure2

Natroba™ (Spinosad) has also demonstrated similar or lower treatment-related adverse events than Nix® (permethrin 1%), the leading OTC medication.6
We have the solution. There is no known resistance to Natroba’s™ active compound, Spinosad.7
Nix® is a registered trademark of Prestige Consumer Healthcare Inc.
Reference(s)
- IQVIA: December 18, 2025.
- Data on File, Cipher Pharmaceuticals Inc.
- Millar, N. S. & Denholm, I. Nicotinic acetylcholine receptors: targets for commercially important insecticides. Invert. Neurosci. 7, 53-66.
- Centers for Disease Control and Prevention (CDC). Clinical Care of Head Lice. Updated January 31, 2025. Accessed January 26, 2026. https://www.cdc.gov/lice/hcp/clinical-care/
- Natroba Prescribing Information
- Stough D, Shellabarger S, Quiring J, Gabrielsen A. Efficacy and safety of spinosad and permethrin crème rinses for pediculosis capitis (head lice). Pediatrics. 2009;124(3): e389-395.
- Spinosad Technical Bulletin Dow AgroSciences LLC (Now Corteva agriscience), Form No. Y45-000-001 (01/01) CBK.
- Gellatly KJ, Krim S,Palenchar DJ, et al. Expansion of the knockdown resistance frequency map for human head lice (phthiraptera: pediculidae) in the United States using quantitative sequencing. Journal of Medical Entomology, 2016:1-7.
NAT-WHL3-000
