It’s Time for a Different Scabies Treatment4
Only Natroba™ (Spinosad) demonstrates the latest clinical and confirmatory outcomes as defined by the FDA’s criteria for "complete cure", and also meets the FDA’s guidance for assessment of safety for topical therapies.1-4
Learn MoreThe need for substantiated results has been unmet for thirty years.
1989
Discretionary Data
In scabies trials, "cured" outcomes were based on observational data1**
2016
New Objective Criteria
FDA puts forth new objective criteria for defining a "complete cure" of scabies2

2020
Guidance For Topical Drugs
FDA issues guidance for topical therapies based on dermal safety studies3

*Oral ivermectin is not FDA-approved for scabies but may be prescribed off-label.
See how Spinosad, the active compound in Natroba™, has a MOA that defines targeted topical therapy5,6
- Not systemically absorbed
- No neurotoxicity
- No documented resistance challenges
Natroba™ (Spinosad) is Superior
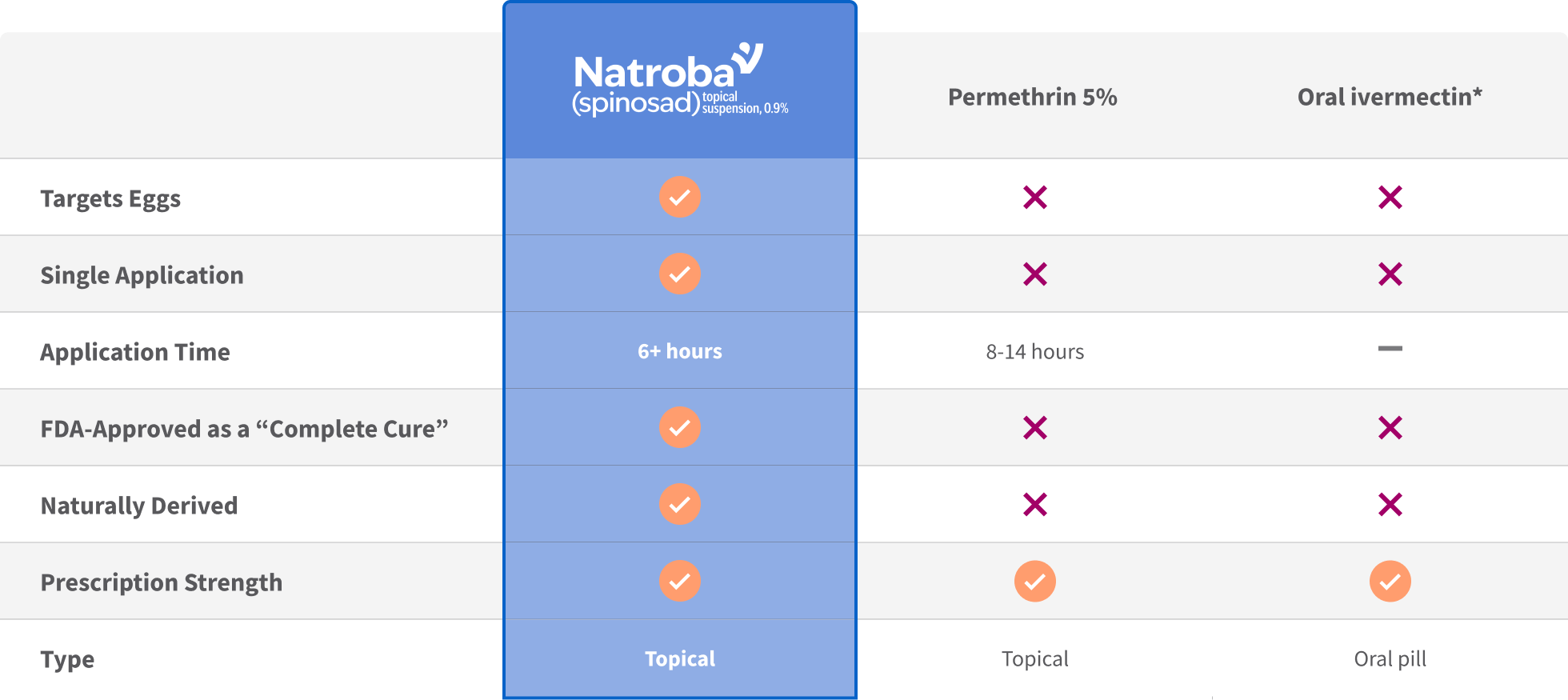
*Oral ivermectin is not FDA-approved for scabies but may be prescribed off-label
- FDA -approved active pharmaceutical ingredient (API), is systemically absorbed and lipophilic7
- Deposits in fat-containing organs (brain, lungs, heart, liver and kidneys)7
- Data suggest that resistance is increasing8-11
- FDA-approved API, and indicated only for second-line treatment12
- Systemically absorbed and lipophilic13-15
- Deposits in fat-containing organs (brain, lungs, heart, liver and kidneys)13-15
- Not FDA-approved API for use with scabies13-15
- Systemically absorbed and lipophilic13-15
- Deposits in fat-containing organs (brain, lungs, heart, liver and kidneys)16
- Data suggest that resistance is increasing17,18
Reference(s)
- Elimite (permethrin) Cream, 5% (NDA 19-855) FDA Approval Leer [Leer wrien August 25, 1989 to Burroughs Wellcome Company].(1989). U.S. Food and Drug Administration. Division of Anti-Infective Drug Products.
- ParaPRO, LLC & Concentrics Research (2018). A Phase 3, Randomized, Double Blind, Placebo-Controlled Study to Assess the Safety, Efficacy and Pharmacokinetics of Natroba™ for the Treatment of Scabies (Clinical Study Protocol, Amd. 6.0, pp. 1-66).
- Center for Drug Evaluation and Research. (2020 March). Contact Dermatitis From Topical Drug Products for Cutaneous Application: Human Safety Assessment Guidance for Industry. U.S. Food and Drug Administration https://www.fda.gov/regulatory-information/search-fda-guidance-documents/contact-dermatitis-topical-drug-products-cutaneous-application-human-safety-assessment-guidance.
- Natroba™ Prescribing Information.
- Data on file, Cipher Pharmaceuticals Inc.
- Spinosad Technical Bulletin Dow AgroSciences LLC (Now Corteva agriscience), Form No. Y45-000-001 (01/01) CBK.
NAT-WHS2-000
